Monitoring and management of ILD progression in CTD-ILDs
Regular monitoring for progression of connective tissue disease-associated interstitial lung diseases (CTD-ILDs) is vital to inform appropriate care1

There is currently no uniformly endorsed definition of the progressive fibrosing phenotype,11 and, in clinical practice, no threshold or rate of decline has been formally accepted12
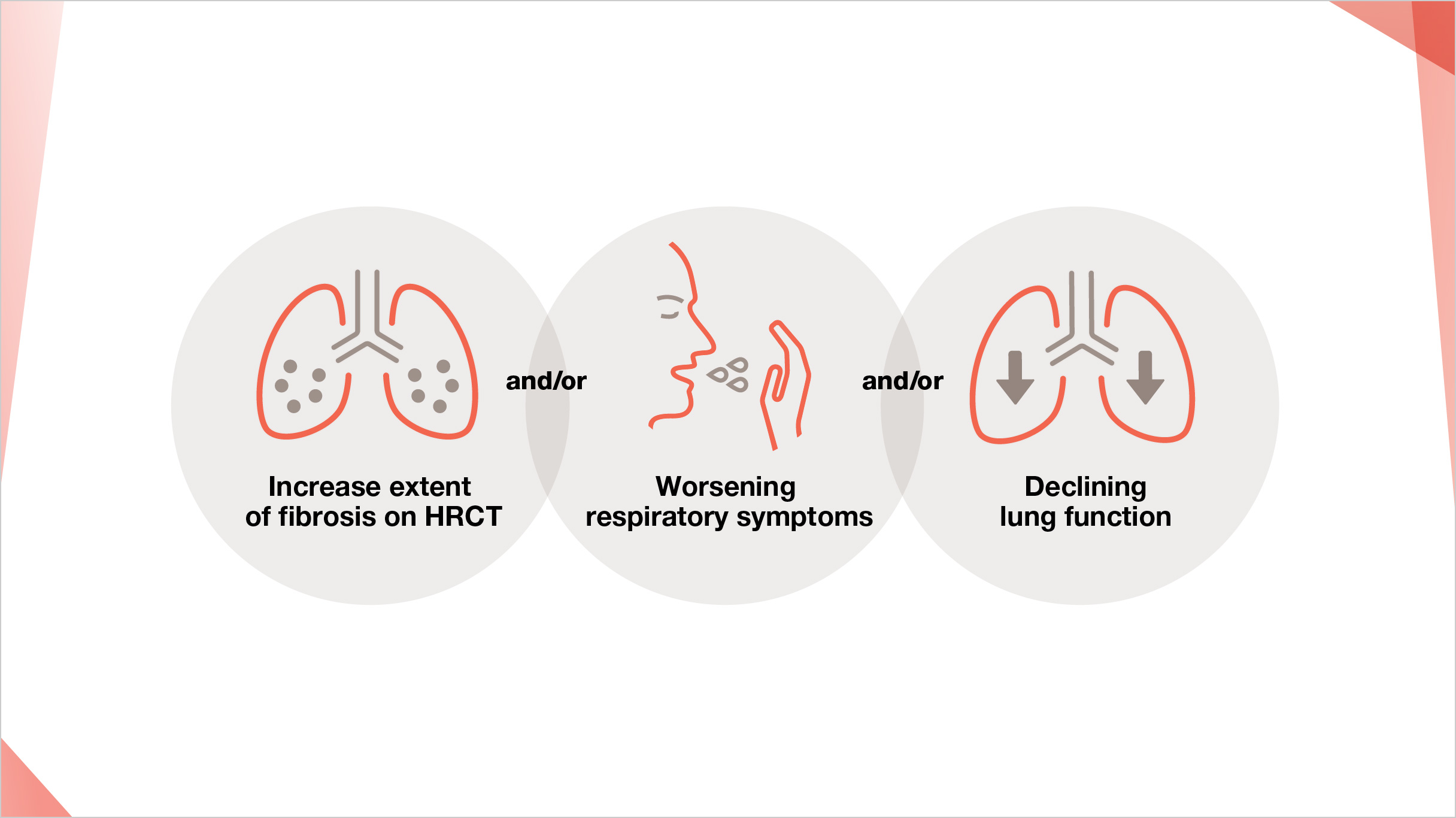
Once alternative explanations, such as respiratory tract infection, have been excluded and treated, patients with ILDs meeting the following criteria at any point within 24 months despite treatment in clinical practice can be considered to have progressive fibrosis:13
-
Relative decline of ≥10% in FVC
-
Relative decline of ≥5–<10% in FVC and relative decline of ≥15% in DLCO
-
Relative decline of ≥5–<10% in FVC and worsening of respiratory symptoms
-
Relative decline of ≥5–<10% in FVC and increased extent of fibrosis on HRCT
-
Worsening of respiratory symptoms and increased extent of fibrosis on HRCT
TIME IS OF THE ESSENCE WHEN PROGRESSIVE FIBROSING ILD IS SUSPECTED9
Delayed detection of progressive pulmonary fibrosis in ILDs can be deadly. Patients have an estimated survival time of 2.5–4 years after detection of progressive fibrosis4,9* Early detection of patients with progressive fibrosing ILD could inform management decisions and patient counselling4
HOW COULD THE TIME BETWEEN ILD DIAGNOSIS AND DETECTION OF THE PROGRESSIVE FIBROSING PHENOTYPE IN YOUR PATIENTS BE SHORTENED?
A patient with CTD-ILD could wait an average of 2.5–3 years from initial ILD symptoms to the diagnosis of progressive fibrosing ILD4*
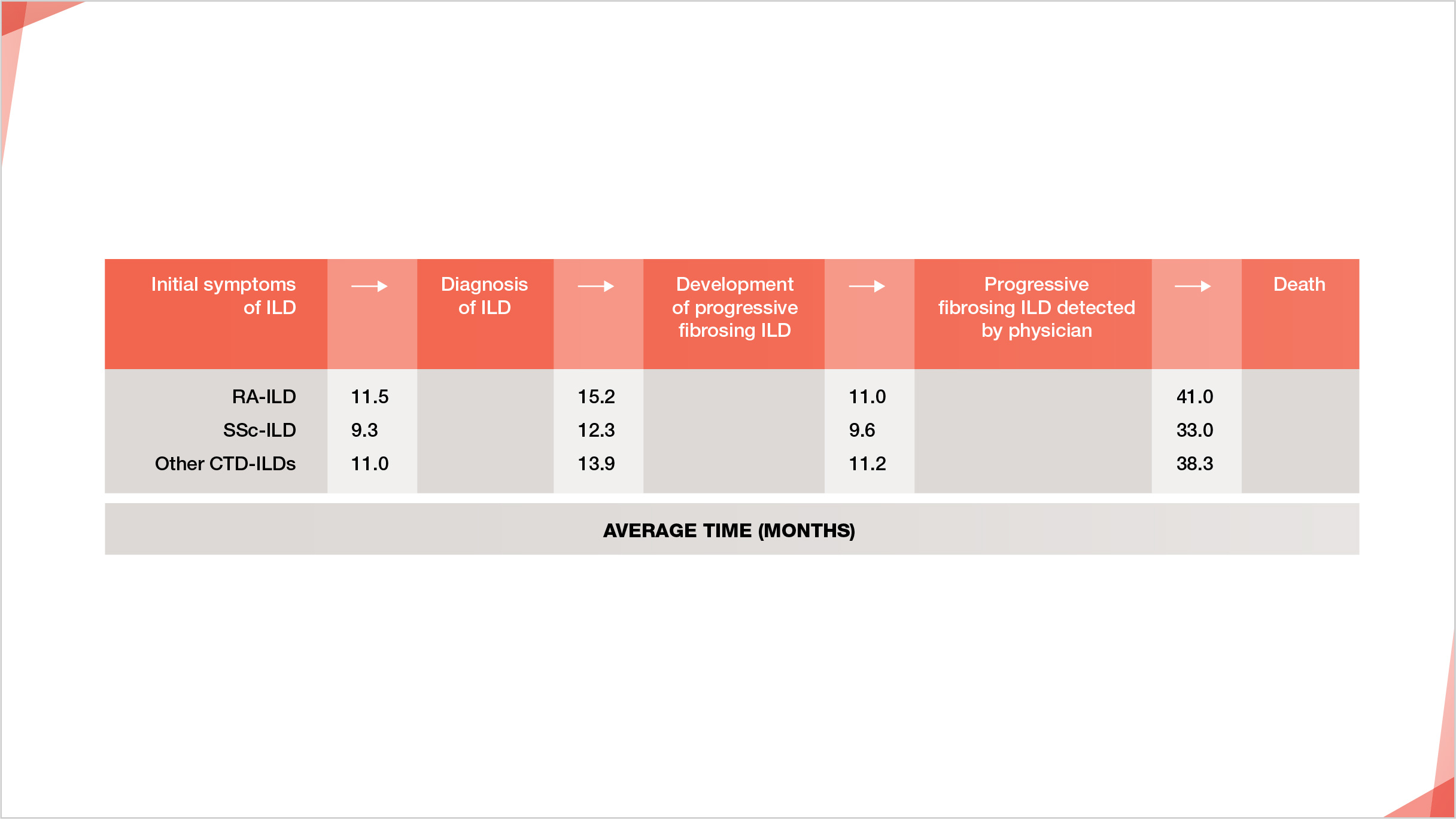
Wijsenbeek M, Kreuter M, Olson A, et al. Progressive fibrosing interstitial lung diseases: current practice in diagnosis and management, 2019, Curr Med Res Opin. Reprinted by permission of the publisher Informa UK Limited trading as Taylor & Francis Ltd.
Patient journey in non-IPF progressive fibrosing ILDs as estimated by an online physician survey*
* Estimates of patients with non-IPF ILD that develop progressive fibrosis based on an online survey of total 486 physicians (243 pulmonologists, 203 rheumatologists and 40 internists) from the United States, Japan, France, Germany, Italy, Spain and the United Kingdom4
IN CTD-ILDs, ILD MONITORING CAN BE OPTIMIZED BY CROSS-SPECIALTY COLLABORATION1
Multidisciplinary evaluation is the gold standard in the diagnosis of ILD and evaluation of its severity14
In instances when an MDT is challenging to convene for a patient with ILD, multidisciplinary discussion can be conducted virtually or by telephone to enable each specialty group to contribute15

Measurements taken during PFTs should include FVC, DLCO and TLC1
Despite its limitations in identifying ILD in patients with CTD, the 6MWD can be used to monitor ILD progression and provide prognostic information for established CTD-ILD – therefore, a regular assessment of the 6MWD is suggested1
Awareness of patient language can help identify the symptoms of ILD in CTDs16

All patients with SSc-ILD need to be followed up every 3–6 months with PFTs, and repeated HRCT as guided by clinical decision, given the highly variable progression of the disease17,18
Learn more about monitoring your patients with SSc-ILD after initiating treatment

Monitoring for RA-ILD progression with PFTs is recommended at 3–6-monthly intervals, then every 6–12 months if stable19
Learn more about monitoring for ILD progression in RA

Monitoring for progression of primary Sjögren's syndrome-ILD using PFTs is recommended every 3–6 months in the first 1–2 years20
Learn more about monitoring for ILD progression in primary Sjögren's syndrome
TREATMENT OF PROGRESSIVE CTD-ILD SHOULD NOT WAIT1,14
A multidisciplinary discussion should be initiated upon diagnosis of ILD in patients with CTD, and during their monitoring, to determine the optimal approach to slow ILD progression12,13
The decision to treat CTD-ILD can be based on whether the patient is clinically impaired by the ILD, whether the ILD is progressive by symptoms, physiology and/or imaging, and what extrathoracic features require therapy1
Proposed algorithm outlining general recommendations for management of ILD in patients with CTD1
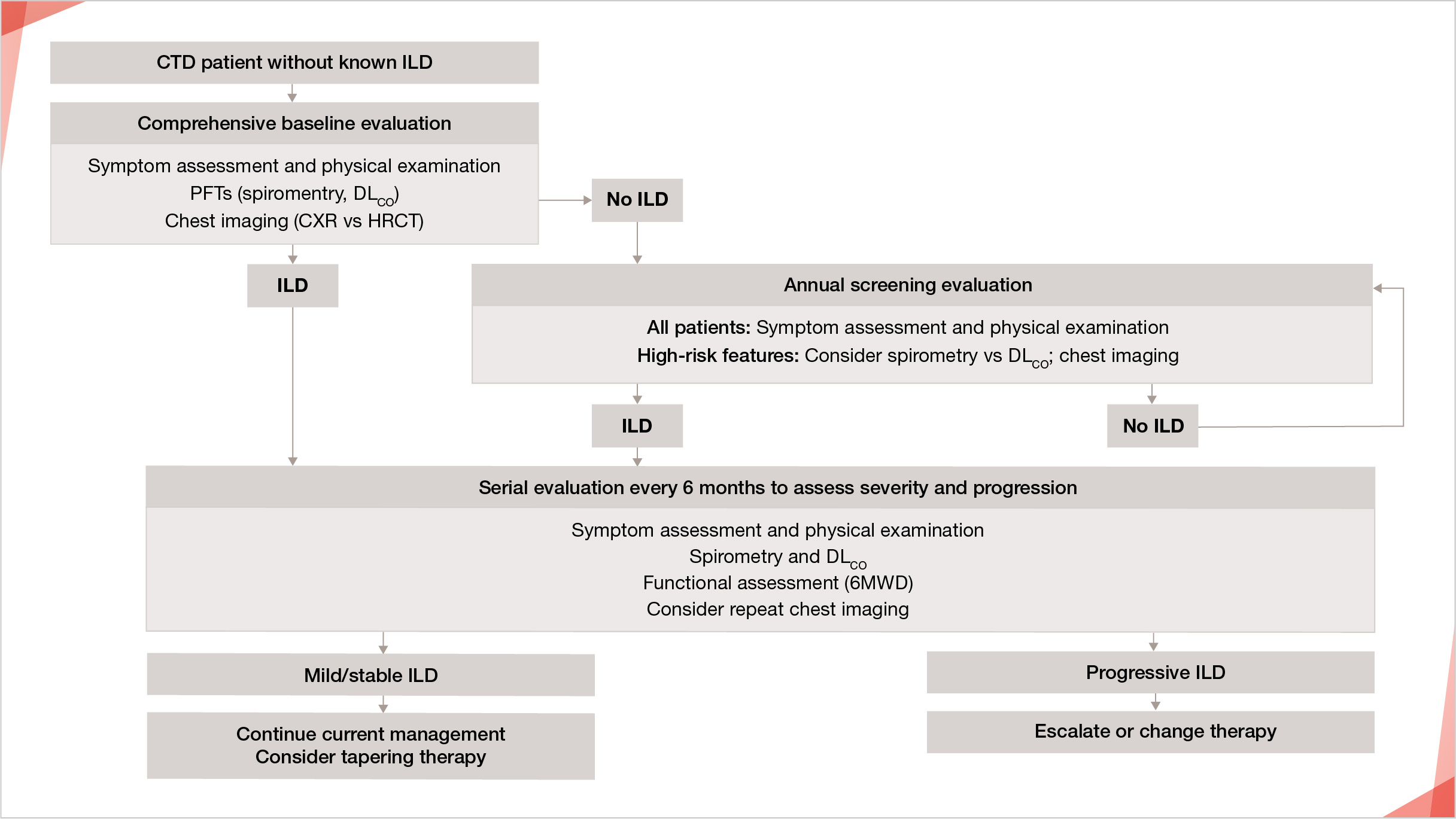
*There is currently no consenus on when to initiate treatment of SSc-ILD.
Adapted from: Geerts S,et al. Sacoidosis Vasc Diffuse Lung Dis. 2017;34:326-335
MANAGEMENT GUIDANCE FOR SPECIFIC CTD-ILDs SHOULD SUPERSEDE GENERAL GUIDANCE FOR MANAGEMENT OF CTD-ILDs AS A WHOLE17,20-23
Some examples are presented below:

Treatment initiation in patients with SSc-ILD should aim for prevention of progression to avoid irreversible lung damage17,21,22
How can you identify and optimize treatment of ILD in your patients with SSc-ILD?

Progression and severity of ILD are the main factors to consider when making treatment decisions for RA-ILD23
How can you identify and optimize treatment of ILD progression in your patients with RA-ILD?

Progression and severity of ILD are the main factors to consider when making treatment decisions for primary Sjögren's syndrome-ILD20
How can you identify and optimize treatment of ILD progression in your patients with primary Sjögren's syndrome-ILD?
How else can you manage your patients with CTD-ILDs?

Management of SSc-ILD

Providing palliative/supportive care

Multidisciplinary teams
Footnotes
-
*
Estimates of patients with non-IPF ILD that develop progressive fibrosis based on an online survey of total 486 physicians (243 pulmonologists, 203 rheumatologists and 40 internists) from the United States, Japan, France, Germany, Italy, Spain and the United Kingdom4
-
†
Depending on the overall prognosis and risks of ILD progression.
-
6MWD, six-minute walking distance; BAL, bronchoalveolar lavage; CTD, connective tissue disease; CTD-ILD, connective tissue disease-associated interstitial lung disease; CXR, chest X-ray; DLCO, diffusing capacity of the lung for carbon monoxide; FVC, forced vital capacity; HRCT, high-resolution computed tomography; HRQoL, health-related quality of life; ILD, interstitial lung disease; IPF, idiopathic pulmonary fibrosis; MDD, multidisciplinary discussion; MDT, multidisciplinary team; PFT, pulmonary function test; RA-ILD, rheumatoid arthritis-associated interstitial lung disease; SSc-ILD, systemic sclerosis-associated interstitial lung disease; TLC, total lung capacity.
-
Geerts S, Wuyts W, De Langhe E, et al. Connective tissue disease associated interstitial pneumonia: a challenge for both rheumatologists and pulmonologists. Sarcoidosis Vasc Diffuse Lung Dis. 2017;34(4):326–335.
-
Cottin V, Wollin L, Fischer A, et al. Fibrosing interstitial lung diseases: knowns and unknowns. Eur Respir Rev. 2019b;28(151):pii:180100.
-
Olson AL, Hartmann N, Padmaja P, et al. Estimation of the Prevalence of Progressive Fibrosing Interstitial Lung Diseases: Systematic Literature Review and Data from a Physician Survey. Adv Ther. 2020; https://doi.org/10.1007/s12325-020-01578-6.
-
Wijsenbeek M, Kreuter M, Olson A, et al. Progressive fibrosing interstitial lung diseases: current practice in diagnosis and management. Curr Med Res Opin. 2019;35(11):2015–2024.
-
Zamora-Legoff JA, Krause ML, Crowson CS, et al. Progressive decline of lung function in rheumatoid arthritis associated interstitial lung disease. Arthritis Rheumatol. 2017;69(3):542–549.
-
Hoffmann-Vold A, Aaløkken TM, Lund MB, et al. Predictive Value of Serial High-Resolution Computed Tomography Analyses and Concurrent Lung Function Tests in Systemic Sclerosis. Arthritis Rheumatol. 2015;67:2205–2212.
-
Marie I, Hatron PY, Dominique S, et al. Short-Term and Long-Term Outcomes of Interstitial Lung Disease in Polymyositis and Dermatomyositis. Arthritis Rheum. 2011;63:3439–3447.
-
Flaherty KR, Brown KK, Wells AU, et al. Design of the PF-ILD trial: A double-blind, randomised, placebo-controlled phase III trial of nintedanib in patients with progressive fibrosing interstitial lung disease. BMJ Open Resp Res. 2017;4(1):e000212.
-
Cottin V, Hirani N, Hotchkin D, et al. Presentation, diagnosis and clinical course of the spectrum of progressive-fibrosing interstitial lung diseases. Eur Respir Rev. 2018;27(150):180076.
-
Kolb M, Vašáková M. The natural history of progressive fibrosing interstitial lung diseases. Respir Res. 2019;20(1).
-
Kolb M, Flaherty KR. The justification for the progressive fibrotic phenotype. Curr Opin Pulm Med 2021; online ahead of print.
-
Wijsenbeek M, Cottin V. Spectrum of Fibrotic Lung Diseases. N Engl J Med. 2020;383:958–968.
-
George PM, Spagnolo P, Kreuter M, et al. Progressive fibrosing interstitial lung disease: clinical uncertainties, consensus recommendations, and research priorities. Lancet Respir Med. 2020;8:925–954.
-
Wells AU, Denton CP. Interstitial lung disease in connective tissue disease— mechanisms and management. Nat Rev Rheumatol. 2014;10(12):728–739.
-
Chung JH, Goldin JG. Interpretation of HRCT Scans in the Diagnosis of IPF: Improving Communication Between Pulmonologists and Radiologists. Lung. 2018;196:561–567.
-
Mittoo S, Frankel S, LeSage D, et al. Patient perspectives in OMERACT provide an anchor for future metric development and improved approaches to healthcare delivery in connective tissue disease related interstitial lung disease (CTD-ILD). Curr Respir Med Rev. 2015;11(2):175–183.
-
Hoffmann-Vold AM, Maher TM, Philpot EE, et al. The identification and management of interstitial lung disease in systemic sclerosis: evidence-based European consensus statements. Lancet Rheum. 2020;2:e71–e83.
-
Distler O, Assassi S, Cottin V, et al. Predictors of progression in systemic sclerosis patients with interstitial lung disease. Eur Respir J. 2020;55;1902026.
-
Iqbal K, Kelly C. Treatment of rheumatoid arthritis-associated interstitial lung disease: a perspective review. Ther Adv Musculoskelet Dis. 2015;7(6):247–267.
-
Lee AS, Scofield RH, Hammitt KM, et al. Consensus guidelines for evaluation and management of pulmonary disease in Sjögren’s. Chest. 2020 Oct 20. doi: 10.1016/j. chest.2020.10.011.
-
Hoffmann-Vold AM, Allanore Y, Alves M, et al. Progressive interstitial lung disease in patients with systemic sclerosis-associated interstitial lung disease in the EUSTAR database. Ann Rheum Dis. 2020a;217455.
-
Distler O, Volkmann ER, Hoffmann-Vold AM, et al. Current and future perspectives on management of systemic sclerosis-associated interstitial lung disease. Expert Rev Clin Immunol. 2019;15;1009–1017.
-
Cassone G, Manfredi A, Vacchi C, et al. Treatment of rheumatoid arthritis-associated interstitial lung disease: lights and shadows. J Clin Med. 2020;9(4):1082. doi: 10.3390/jcm9041082.
-
Ryerson CJ, Cayou C, Topp F, et al. Pulmonary rehabilitation improves long-term outcomes in interstitial lung disease: a prospective cohort study. Respir Med. 2014;108(1):203-210.
-
Kreuter M, Bendstrup E, Russell A, et al. Palliative care in interstitial lung disease: living well. Lancet Respir Med. 2017;5(12):968-980.
-
Maher TM, Wuyts W. Management of Fibrosing Interstitial Lung Diseases. Adv Ther. 2019;doi:10.1007/s12325-019-00992-9. [Epub ahead of print].
-
Sgalla G, Cerri S, Ferrari R, et al. Mindfulness-based stress reduction in patients with interstitial lung diseases: a pilot, single-centre observational study on safety and efficacy. BMJ Open Respir Res. 2015;2(1):e000065.
Resources for patients




